The process and its advantages
Nitriding (or nitridation) enriches workpieces with nitrogen, causing nitrides to form within them. The nitrogen penetrates the workpiece and forms on its surface a hard compound layer composed of iron nitrides. Located underneath is the diffusion zone. In this zone, nitrogen embeds in the metal matrix of the workpiece, thus increasing its fatigue strength. The highest values can be achieved with nitriding steels.
Nitriding requires temperatures between 500°C and 580°C. Treatment times vary between one and one hundred hours. The thickness of the compound layer and the development of the porous zones depend on the temperature and duration of the treatment. To define the nitriding hardness depth (NHD) of the treatment, the limit hardness is used as a reference. This is 50 HV higher than the core hardness of the workpiece.
The layer’s resistance to corrosion can be further improved by oxidation. If additional coatings (galvanic or chemical) are planned later on, the workpiece can also be nitrided without a compound layer.
The process is different from nitrocarburising, which involves the additional use of carbon.
Graphical representation: Structure and functional principle
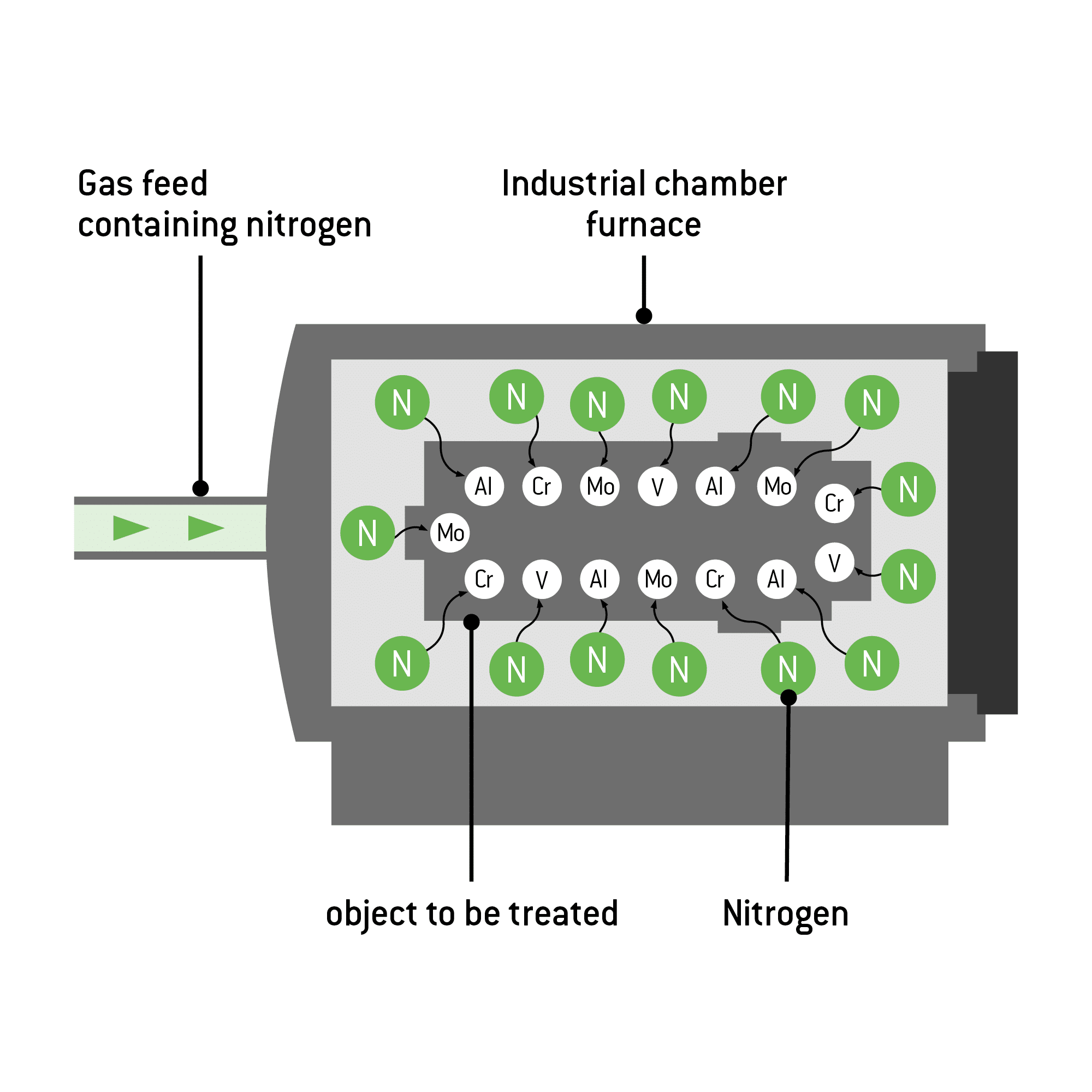
The most commonly used nitriding processes are:
Each of these processes also permits partial nitriding. During salt bath nitriding, the workpieces are dipped only partially in the salt bath. The partial treatment during plasma nitriding is accomplished by applying a protective compound or by mechanical means.
The advantages of nitriding at a glance
- Improved hardness of the surface
- Less wear
- High resistance to corrosion
- Lower friction coefficient
- The nitrided layer is heat-resistant up to 600°C
- Partial hardening possible
- Many possible applications, because almost any type of steel can be nitrided. Suited best, however, are alloy steels.
Areas of application: Suitable steels
Almost all cast and sintered materials and grades of steel can be nitrided.
Alloy steels achieve better results than non-alloy steels. With the latter, the surface of the material may become brittle instead of hard. For this reason, low- or non-alloy steels are nitrocarburised instead. This process produces a compound layer that improves protection against wear and corrosion. This compound layer can also be subjected to post-oxidation. That creates another thin oxide layer with a thickness between 1 – 3 µm and provides even greater protection.
In contrast, elements such as molybdenum, titanium, chromium, or aluminium contained in alloy steels produce particularly hard nitrides when reacting with nitrogen. Steels containing a particularly high content of these elements are therefore also referred to as nitriding steels.
These include:
- 1.8519 (31CrMoV9)
- 1.8515 (31CrMo12)
- 1.8550 (34CrAINi7)
- 1.8507 (34CrAlMo5)
You can find a comparison of different methods in the table below.
Process locations
We provide state-of-the-art nitriding processes at the following locations: location overview
Check list: Ordering nitriding
If you would like us to nitride workpieces or materials, we will gladly advise you on the best procedure. You can use the check list below to make your preparations for the order.
- Which material is to be treated, and what is its condition?
- What is the target hardness (including tolerance range in HV)?
- What is the desired nitriding hardness depth (including tolerance range in mm)?
- If applicable, which areas should be nitrided, and where can the hardness be measured?
- If applicable, how thick should the compound layer be (including tolerance range in μm)?
Note: We use a sample that we take ourselves to measure the thickness of the compound layer and/or the nitriding hardness depth. For measurements that are specifically related to your order, we need you to provide us with a reference component that is intended for the hardening treatment.